Guidelines:
|
|
|
|
|
- Table 1: Sample Quantity Requirements
| Potency*/per active | Microbiology | Other Tests | ||||
|
|
|
|
||||
| Aliquots / Powders | 1 gram*** | Sterility** | 2 mL | pH | 15 mL | |
|
|
|
|
||||
| Capsules, Tablets, Pellets | 5 or more | Endotoxin | 1 mL | Specific Gravity | 30 mL | |
|
|
|
|||||
| Liquid Samples | 5 mL | Particulates USP <788> SVI (Method 1 and 2) |
For fill ≥ 25 mL, 1 container For fill 1 mL to 25 mL, 10 containers For fill < 1mL, 10 mL |
|||
|
|
|
|
||||
| Suppositories, Troches, Lollipops | 5 or more | Particulates USP <788> LVI (Method 1 and 2) |
For fill > 100 mL, 1 container | |||
|
|
|
|
||||
| Creams, Lotions, Gels | 5 grams | Particulates (Visual) USP <797> | 1 mL | |||
|
|
|
|
||||
| Antimicrobial Effectiveness | 100 mL | |||||
|
|
|
|
||||
| * Per active, per time point.
** See USP <71> (Table 3) for sampling guidelines to meet USP requirements *** If testing Levothyroxine Sodium (T4) and Liothyronine (T3) please submit 5 grams |
||||||
| Notes: Please send separate sample containers for each test requested. If testing multiple sterility tests (i.e.: in the event of a time study), please send individual containers for each time point. | ||||||
Table 2: Pricing and Turnaround Times (TAT) Service Levels
| Standard Test Turnaround Times (Routine Testing)
✓ Chemical/Physical Test Final Report/CoA
✓ Microbiology
Rush Pricing ✓ Same Business Day: Call for Fee (some compounds/actives cannot be tested Same Business Day) ✓ Next Business Day / Same day setup for Sterility: 2X Standard Pricing per test ✓ 2nd Business Day / Next business day setup for Sterility: 1.5X Standard Pricing per test Project and Service Levels
|
CLIENT REQUIREMENTS FOR SERVICE LEVEL COMPLIANCE
- Applies to Compounds currently validated by DYNALABS – New Compounds Need to be Fully Technically Veted prior to being included in this Service Level Agreement (SLA)
- No Shared Sample (minimum 2 – 5 ml per test)
- “Day 0” is the date the sample is received on our systems (De-Boxing, Sample Verification, etc.), not necessarily the date of delivery
- Required Information Provided from Pharmacy must mesh with DYNALABS’ systems – Test Method and Specifications Available, Sample Information is complete and accurate (Formulation, Lot Number, Sample Description, Volume, Batch Size, Sample Identifiers, etc.). Incomplete data will result in delays. Project Information, e.g. approved quote, type of project, etc.
- For Rush Samples, identified as “rush” during time of submission
• Same Day – Call for Fee and Capability
• Samples On Site by 10 AM CST
• Sample Request Submitted by 7 AM CST - Investigations and/or Issues will proceed per Standard Operating Procedure (SOP) and not Service Levels
Table 3: Minimum numbers of articles to be tested in relation to the numbers of articles in the batch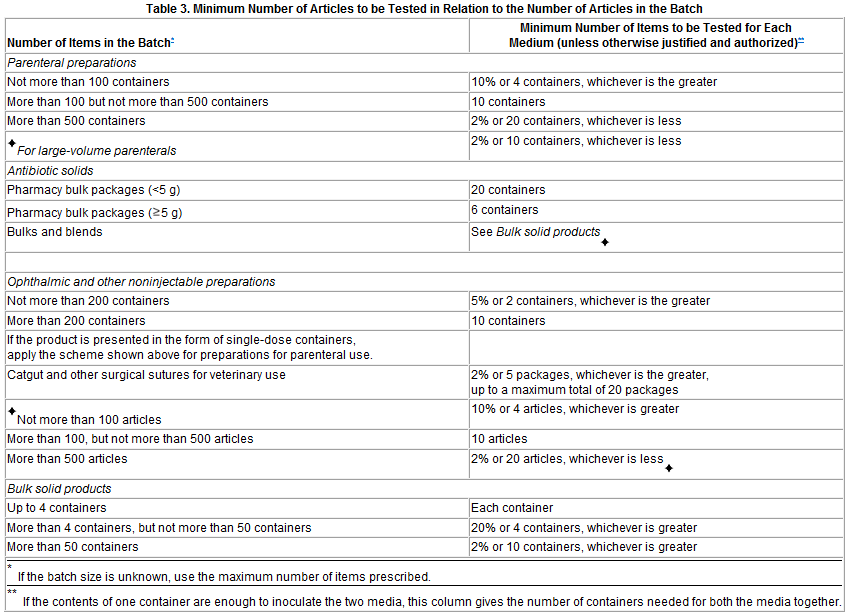
Confidentiality
DYNALABS maintains strict confidentiality with its customers. Formal Confidentiality agreements may be initiated by the customer or by DYNALABS.
Retention and Disposal
Samples will be retained 8 days after final QA. Prior arrangements must be made to retain samples under other conditions or to return samples. Raw data is retained per cGMP standards and to maintain 21 CFR compliance.
Controlled Substance – 222 Form
DYNALABS possesses a DEA license for Schedules II – V controlled substances.
A DEA 222 form is not always required each time you send in a controlled substance.
TITLE 21 — FOOD and DRUGS
CHAPTER II — DRUG ENFORCEMENT ADMINISTRATION
DEPARTMENT OF JUSTICE
PART 1305 — ORDERS FOR SCHEDULE I AND II CONTROLLED SUBSTANCES
Subpart A — General Requirements
Sec. 1305.03 Distributions requiring a Form 222 or a digitally signed electronic order.
Either a DEA Form 222 or its electronic equivalent as set forth in subpart C of this part and Part 1311 of this chapter is required for each distribution of a Schedule I or II controlled substance except for the following:
- Distributions to persons exempted from registration under Part 1301 of this chapter.
- Exports from the United States that conform to the requirements of the Act.
- Deliveries to a registered analytical laboratory or its agent approved by DEA.*
- Delivery from a central fill pharmacy, as defined in 1300.01 (b) (44) of this chapter, to a retail pharmacy.
* NB: Drug manufacturers, drug repackagers and drug distributors are required to complete and submit a DEA Form 222 or its electronic equivalent when sending Schedule I and II controlled substances for testing. Please contact us regarding any DEA Form 222 requirements questions you may have.
Financial Information
- Payment in advance may be required for customers whose credit has not been established with DYNALABS.
- Purchase orders or valid credit card information are required, unless other credit options have been approved by DYNALABS. Contracts for Services without this information may cause delay in testing.
- Payment terms are “Net 15” from date of invoice unless otherwise stated differently in a quote. Client agrees to pay all costs, including, but not limited to, attorney and accounting fees and other expenses of collection resulting from any default by client under any terms of this contract.
- For third party billing, a signed purchase order must be received from the party being billed for the service.
- Published pricing is designed for single samples. Project or volume discounts may be available.
- Quotations will be provided upon request by the client.
- Additional charges may be assessed (i.e. hazardous samples) with client approval.
- Liability of DYNALABS is limited to an amount no greater than the amount invoiced. This document supersedes any other arrangements with regards to financial liability.

