 You Can Trust the Result
You Can Trust the Result
We use an FDA compliant Outside of Specification (OOS) process to investigate all OOS test results before they are reported. Guidance for Industry: Investigating Out-Of-Specification (OOS) Test Results for Pharmaceutical Production-U.S Department of Health and Human Service Food and Drug Administration Center for Drug Evaluation and Research (CDER), October 2006 Pharmaceutical cGMPs and Pharmaceutical Quality Control Labs (7/93)-Guide to Inspections of Pharmaceutical Quality Control Laboratories are the resources used to guide our investigations and are cited throughout our OOS Department SOPs. Please feel free to ask us for a copy!
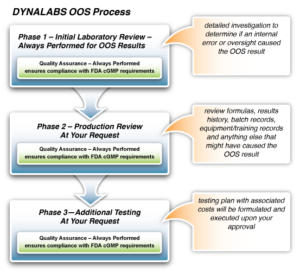
Before we notify you of an OOS result, we perform an internal review of anything DYNALABS might have done to contribute to the OOS result. All OOS investigations include a Phase I and Phase II review before a customer is ever notified of an OOS result. The two-part review not only follows regulatory guidance but ensures an internal error has not been identified.
The initial review, performed by laboratory leads and SMEs, includes:
- Verification of sample preparation and dilution calculations per our internal processes. This includes verification of the glassware used in the preparation.
- Verification of sample label
- Verification of method and instrument used during the sample run
- Verification of regents and possible environmental factors that may have contributed to the OOS
The secondary review, performed by OOS Investigation Analysts, includes:
- Thorough review of data packet and associated paperwork
- Thorough review of Phase I information
- Review of sample submission which may include review of formula
- Any additional investigational work needed such as but not limited to further analysis of chromatography by PDA or visual verification of turbidity.
Only after we have thoroughly reviewed the data, methods, equipment, storage conditions through initial and secondary review and have not identified an internal error, do we contact you with the OOS result.
You Can Find Out What Happened – Just Ask
We are glad to work with you to find out what caused the OOS result and determine how to prevent it in the future.
Our collaborative approach relies on your cooperation to review formulas, results history, batch records, equipment/training records and anything else that might have contributed to the failure.
We will use our expert knowledge of OOS investigations, sample preparation, and testing to help you:
- Discover issues and aid in finding solutions to prevent similar OOS results in the future
- Run additional tests to ensure the implemented solutions perform as expected. This may include work inside an investigation or quoted projects.
In addition, DYNALABS offers consultant services that can aid in batch record review and revisions, process evaluations and audits that may aid manufacturing facilities in preventing OOS and compliance with guidance.
Still Have Questions? Come Visit Us
DYNALABS is pleased to host you for a Quality Systems audit. Please contact your Sales Representative, if you would like to visit our facility. We ask only that you give us at least 30 days of notice.
DYNALABS is dedicated to providing solutions, not just results, and we have the certifications and reputation to back it up:
- FDA registered
- Quality systems meeting cGMP (Current Good Manufacturing Practices) Regulations
- ISO 17025 accredited
- DEA Licensed
- Bureau of Narcotics and Dangerous Drugs Licensed in Missouri
- Protected by U.S. Patent Numbers 7,197,405 and 7,660,678

